Coronavirus qRT PCR Detection Kit

Sentebiolab Senteligo Coronavirus (COVID-19) SARS CoV-2 Multiplex qPCR Test Kit
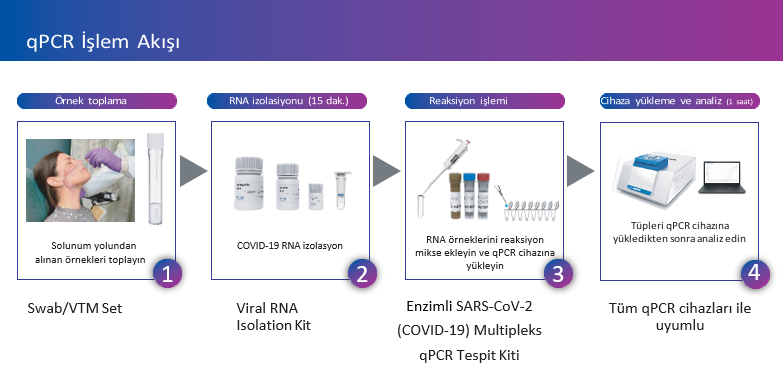
As Sentebiolab, we have been producing innovative and high-tech solutions to control the spread of the virus since the beginning of the pandemic with our products. Our laboratory has developed the Coronavirus Test Kit – enzyme “SenteligoTM SARS – CoV – 2 (Covid-19) Multiplex qPCR Detection Kits”. Along with our diagnostic kit, we have a set that includes a complementary RNA isolation kit, nasopharyngeal swab and viral tubes. Along with our products, we offer our customers a qPCR device upon request and design and install a fully-equipped COVID-19 test laboratory. You can contact us for our references.
FEATURES
Fast results and ease of use
High reliability (99%)
Acceptance worldwide
Minimized human error & contamination risk
High sensitivity and specificity
ADVANTAGES
Primer/probe sequence optimized to minimize false-positive results
Internal Control
• RNA quality control to minimize false-negative results
• Control that enzymes and other reagents are working correctly
100% recognition of unmutated SARS-CoV-2 target regions thanks to Primer-Probe sequence
Sentebiolab laboratories have all the necessary quality documents and conformity assessments with the approval of the Ministry of Health. We produce 500,000 coronavirus test kits weekly in our laboratory with full technical and professional staff.